Prof. Zhi-Kuang Tan, Jax Lee
Organic light-emitting diodes (OLED) and quantum dot enhanced liquid crystal displays (QD-LCD, advertised by Samsung as QLED) have successfully achieved commercial production after two decades of intense research and development. OLED functions through electroluminescence and relies on the direct injection of charges into organic emitters, typically vacuum-sublimation-deposited, to achieve light emission (see Figure 1a). The attainment of display images via the on-and-off switching of individual pixels have been touted by display manufacturers to provide the best contrast given that any pixels turned off would naturally be completely black. Such mode of operation is also expected to offer higher power efficiency – unlike LCD displays that operate by blocking light, OLED displays switch individual pixels on only when required. While there are multiple merits associated with OLED displays, they also suffer a significant shortcoming in colour quality. This is due to the fact that organic semiconductors possess broad excitonic emissions peaks with spectral widths that are often in excess of 100 nm, which limits the amount of colours the display can accurately produce. This problem is solved to a certain extent through the use of spectral-shaping colour filters, but this negates the advantage of power efficiency.
QD-LCD, on the other hand, employs a blue backlight-unit and obtains red and green emissions through a photoluminescent colour-enhancement film comprising of quantum dots. The combined red, green and blue (RGB) backlight then passes through an optical film stack and a liquid crystal panel with RGB colour filters to form the display image (see Figure 1b). Here, the orientation of the liquid crystals is switched with an applied field, which then allows varying amounts of polarized backlight to exit the display and form an image. The QDs function only at the backlight portion of the display to provide spectrally-sharper green and red emission. The sharper spectra, in turn, is capable of providing a wider range of colour sensations for the human eyes as the RGB cone cells in our eyes’ retina can now be more-selectively triggered by the display. This also means that the QD-LCD display is able to reproduce colours more accurately based on the scene where the image was taken given its ability to produce a wider gamut of colours.
(a)
(b)
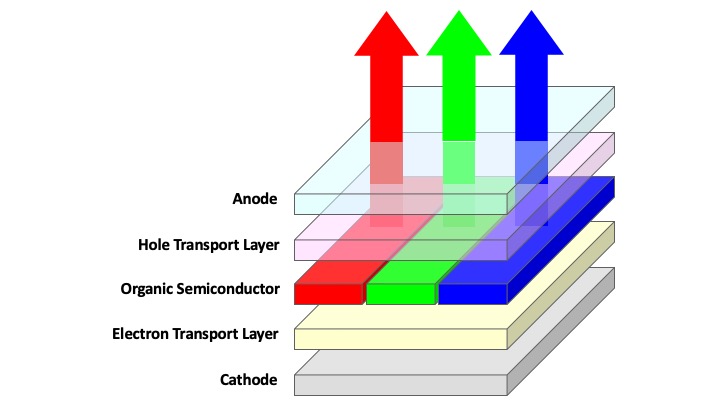
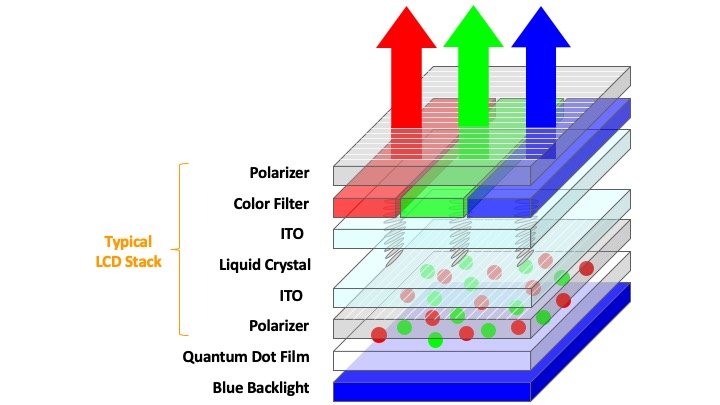
Figure 1. (a) Device configuration of a typical OLED display. (b) Device configuration of a typical QD-LCD display.
Despite the promise of colour enhancement by quantum dots in QD-LCDs, their colour performance remains non-ideal and leaves significant room for improvements. This is primarily because many QD-LCD displays employ Indium Phosphide (InP) quantum dots as their red and green emitters, which possess rather broad spectral width of 40-50 nm. Quantum dots that are based on Cadmium Selenide (CdSe) are capable of achieving narrower spectral width of 20-30 nm, but are heavily-regulated with the risk of being banned in many countries due to the high toxicity of cadmium. In particular, RoHS regulations require consumer electronics products to contain no more than 100 ppm of Cd, which severely limits their use in QD-LCD. As a result, non-Cd-based QDs such as InP have gained commercial adoption, but offer considerably poorer colour performance.
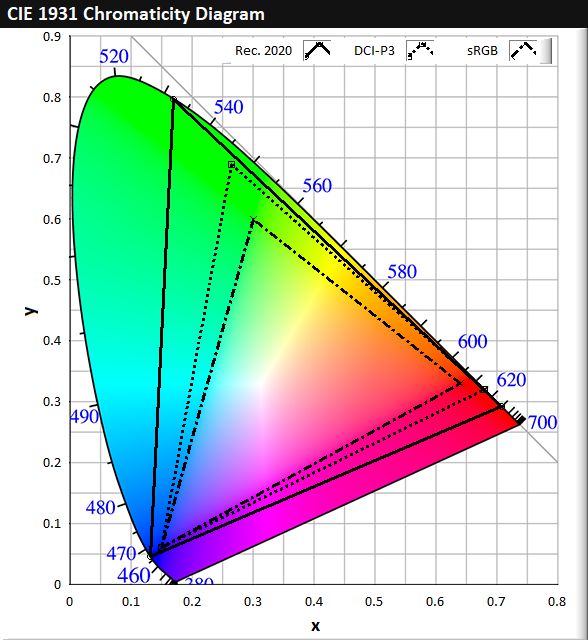
Figure 2a is a CIE 1931 colour space chromaticity diagram, which is a graphical representation of all the colours that can be seen by the average human eye. The triangles within the diagram are three common colour space standards that are used in the display industry and represent the colours that the display is capable of fulfilling. The vertices of the triangles are formed by red, green and blue colours, and the colours within the triangles are formed by a combination of RGB at varying ratios. sRGB is the most basic colour standard with the smallest colour space, and almost all modern displays are capable of fulfilling this standard. Higher-end displays such as OLEDs or QD-LCDs are generally capable of fulfilling only the DCI-P3 standard. For instance, Figure 2b shows a comparison of the colour space coverage by the latest 2019 models of LG OLED C9 and Samsung QLED Q900R televisions with DCI-P3.
(a)
(b)
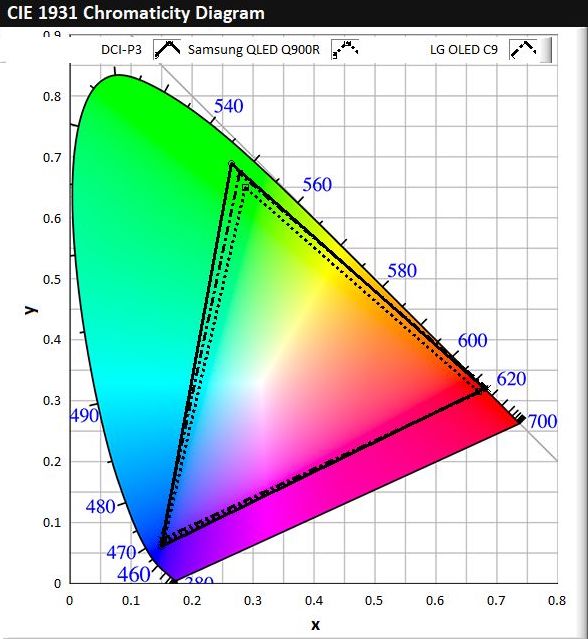
Figure 2. (a) CIE 1931 colour space chromaticity diagram, showing colour space of sRGB, DCI-P3 and Rec. 2020 standards. (b) Comparison of colour space coverage between 2019 models of Samsung QLED Q900R and LG OLED C9 televisions with DCI-P3.
However, it is clear that a considerable fraction of the observable and nature occurring colours are still missing from the DCI-P3 standard. Therefore, there is a significant demand to move towards the Rec. 2020 colour standard, which encompasses 100% of the Pointer’s gamut (natural occurring colours), bringing absolute realism to displays. Displays that can fulfill the Rec. 2020 standard are capable of producing more realistic colours, which remarkably enhances the viewing experience for televisions, computer monitors, smartphones, tablets or advanced tech gadgets such as augmented reality or virtual reality devices. There are, however, no competitive solutions available for achieving Rec. 2020 standard, not unless the light-emitters employ toxic cadmium.
A new semiconductor based on lead halide perovskites (e.g. CsPbBr3 and MAPbI3) has recently emerged as a highly-promising candidate towards fulfilling the Rec. 2020 standard in colour displays.
In 2014, Prof Tan Zhi Kuang, co-founder of Nanolumi, discovered that perovskite semiconductors are capable of exhibiting strong luminescence and successfully developed the first functional perovskite-based light-emitting diodes (Z. K. Tan et al. Nature Nanotechnology 9, 687).1 In this early work, perovskite was shown to be capable of emitting light in a variety of colours through the tailoring of its halide composition, and electroluminescence in the near-infrared, green and red were successfully demonstrated.
In early 2015, Prof Tan and coworkers further reported the formation of perovskite nanocrystals through the blending of perovskite precursors within an organic matrix, and successfully demonstrated wavelength tuning through size control2 as well as showed strong electroluminescence from the nanocrystal-based devices.3 At around the same time, the group of Kovalenko reported a convenient chemical synthesis method to produce colloidal perovskite nanocrystals that can be tuned to emit across the entire visible spectrum at remarkably high efficiencies.4 In early 2016, Prof Tan and coworkers employed colloidal nanocrystals in LEDs and reported high electroluminescence quantum efficiencies of 5.7%, which was then the champion LED efficiency amongst other reports (see Figure 3a).5 Further research efforts by several groups were successful in pushing device efficiencies in excess of 20%.6-8 Last year, Prof Tan’s group demonstrated highly-efficient large-area devices in both rigid and flexible form-factors (Figure 3b) (X. Zhao. & Z.-K. Tan et al. Nature Photonics).8
(a)

(b)

Figure 3. (a) Photos of colourful light-emitting diodes based on colloidal perovskite nanocrystals. (b) Flexible large-area perovskite light-emitting diodes.
The interest in perovskite semiconductors for displays stem primarily from its remarkably high luminescence efficiency (often close to unity), its narrow spectral width (15–30nm), as well as its ability to be tuned to emit in practically all colours spanning the entire visible spectrum. Figure 4a shows perovskite semiconductors that are perfectly-tuned to emit in RGB at 640nm, 530nm, and 460nm. The corresponding coverage of the CIE 1931 colour space is displayed in Figure 4b and shows a > 95% overlap with the Rec. 2020 standards. Such qualities allow perovskites to be highly competitive with Cd-based quantum dots in nearly all aspects of optical performance. Although current high efficiency perovskite semiconductors often contain lead (Pb), its toxicity is significantly lower than Cd, and has a 10-times higher RoHS usage limit of 1,000 ppm. This represents a unique opportunity for perovskites and the use of it as a spectrally-sharp emitter in the next generation of advanced colour displays.
(a)
(b)
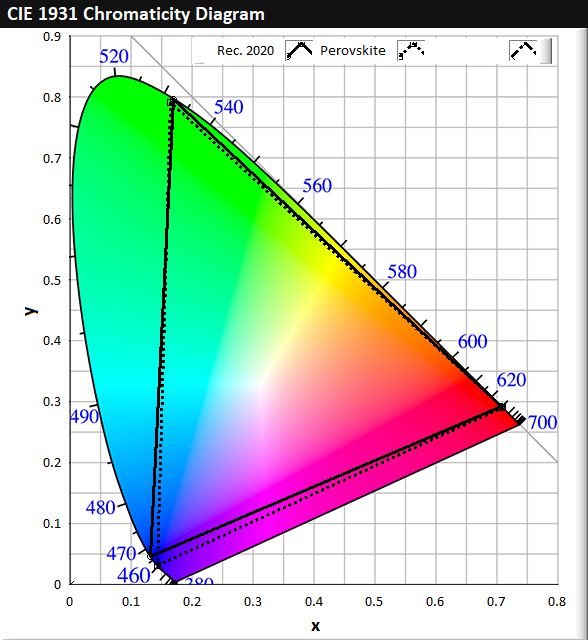
Figure 4. (a) Luminescence spectra of perovskite semiconductors tuned to emit at 460, 530 and 640nm. (b) CIE 1931 colour space chromaticity diagram, showing colour space coverage of perovskite emission compared against Rec. 2020.
Unlike other core-shell quantum dots, perovskite nanocrystals do not usually possess an outer-shell for protection. This could make them more susceptible to attack by ambient molecular species such as water or oxygen, although literature reports have suggested that perovskites are exceptionally defect-tolerant due to their shallow defect states. Nanolumi has successfully engineered strong ligand shells that possess remarkably strong affinity to the perovskite surface and thus effectively strengthen the stability of the nanocrystals. Nanolumi’s researchers have also shown in earlier works that a dense protective polymeric shell could be built around perovskite nanocrystals via a self-initiated photo-polymerization reaction with olefins (e.g. styrene).9 The polymeric shells were effective in encapsulating the crystals and in repelling water from the nanocrystal surface. More crucially, these polymeric shells serve to keep the nanocrystals spatially-separated and prevent them from fusing into larger crystallites when in close proximity, thus offering enhanced stability at elevated temperatures.
The robustness of Nanolumi’s perovskite materials and applications were validated under stringent industry-compliant reliability studies, involving high heat, high humidity, and high light irradiation conditions. More specifically, we test samples in heated chambers at 85°C for heat tests, in humidity chambers at 60°C / 90%RH for humidity tests, and under greater than 100 mW/cm2 blue light irradiance for light irradiation tests. Perovskites that were engineered with strong ligand shells and polymeric composites have demonstrated remarkable stability against these test conditions.
An ideal combination of excellent colour performance, high quantum efficiencies, facile synthesis, low toxicity and robust stability makes perovskite semiconductors the most promising advanced material for the next-generation of ultra-high definition colour displays. Based on our company’s rapid development trajectory, we envision the roadmap for perovskites in displays to start with colour enhancement films, followed closely by colour conversion layers in OLEDs and microLEDs, and eventually making an impact in full-fledged electroluminescent perovskite displays.
References
- Tan, Z.-K. et al. Bright light-emitting diodes based on organometal halide perovskite. Nature Nanotechnology 9, 687 (2014).
- Di, D. et al. Size-Dependent Photon Emission from Organometal Halide Perovskite Nanocrystals Embedded in an Organic Matrix. The Journal of Physical Chemistry Letters 6, 446-450 (2015).
- Li, G. et al. Efficient Light-Emitting Diodes Based on Nanocrystalline Perovskite in a Dielectric Polymer Matrix. Nano Letters 15, 2640-2644 (2015).
- Protesescu, L. et al. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Letters 15, 3692-3696 (2015).
- Li, G. et al. Highly Efficient Perovskite Nanocrystal Light-Emitting Diodes Enabled by a Universal Crosslinking Method. Advanced Materials 28, 3528-3534 (2016).
- Lin, K. et al. Perovskite light-emitting diodes with external quantum efficiency exceeding 20 per cent. Nature 562, 245-248 (2018).
- Cao, Y. et al. Perovskite light-emitting diodes based on spontaneously formed submicrometre-scale structures. Nature 562, 249-253 (2018).
- Zhao, X. & Tan, Z.-K. Large-area near-infrared perovskite light-emitting diodes. Nature Photonics (2019).
- Wong, Y.-C., De Andrew Ng, J. & Tan, Z.-K. Perovskite-Initiated Photopolymerization for Singly Dispersed Luminescent Nanocomposites. Advanced Materials 30, 1800774 (2018).


